Do Metals Form Anions Or Cations
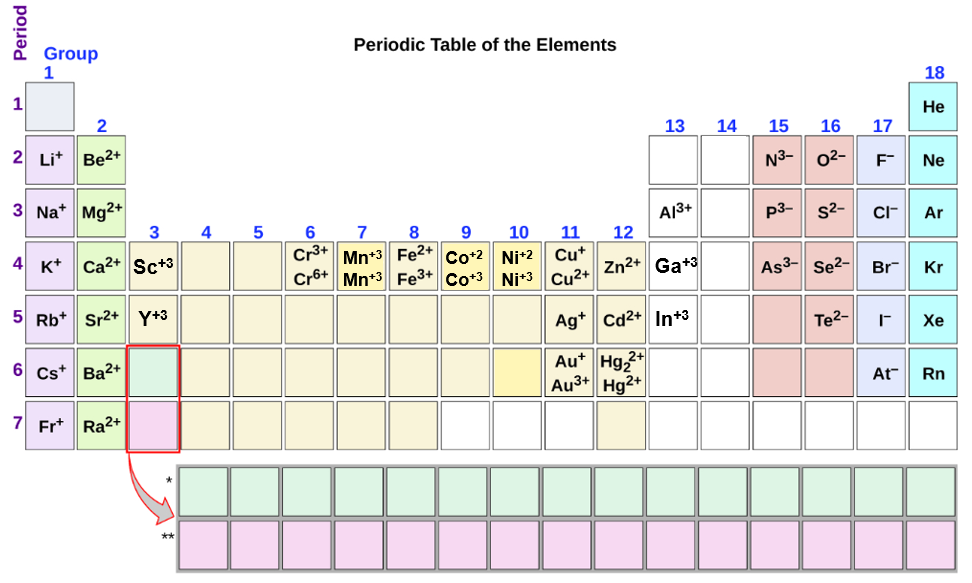
Do Metals Form Anions Or Cations - Web most transition metals differ from the metals of groups 1, 2, and 13 in that they are capable of forming more than one cation with different ionic charges. Nonmetals will form anions, except for the noble gases,. Loses 1 electron to form 1+ ion. Web tara_donoghue terms in this set (60) what distinguishes a neutral atom from an ion ion has a different number of electrons do nonmetals form anions or cations? Web many elements can take the form of either anions or cations depending on the situation. Web cations are positively charged ions. Web what do metals form? Anions are negatively charged ions. These ions are negative because they contain more electrons than protons. Web atoms gain electrons in their outer shell when they form negative ions, called anions.
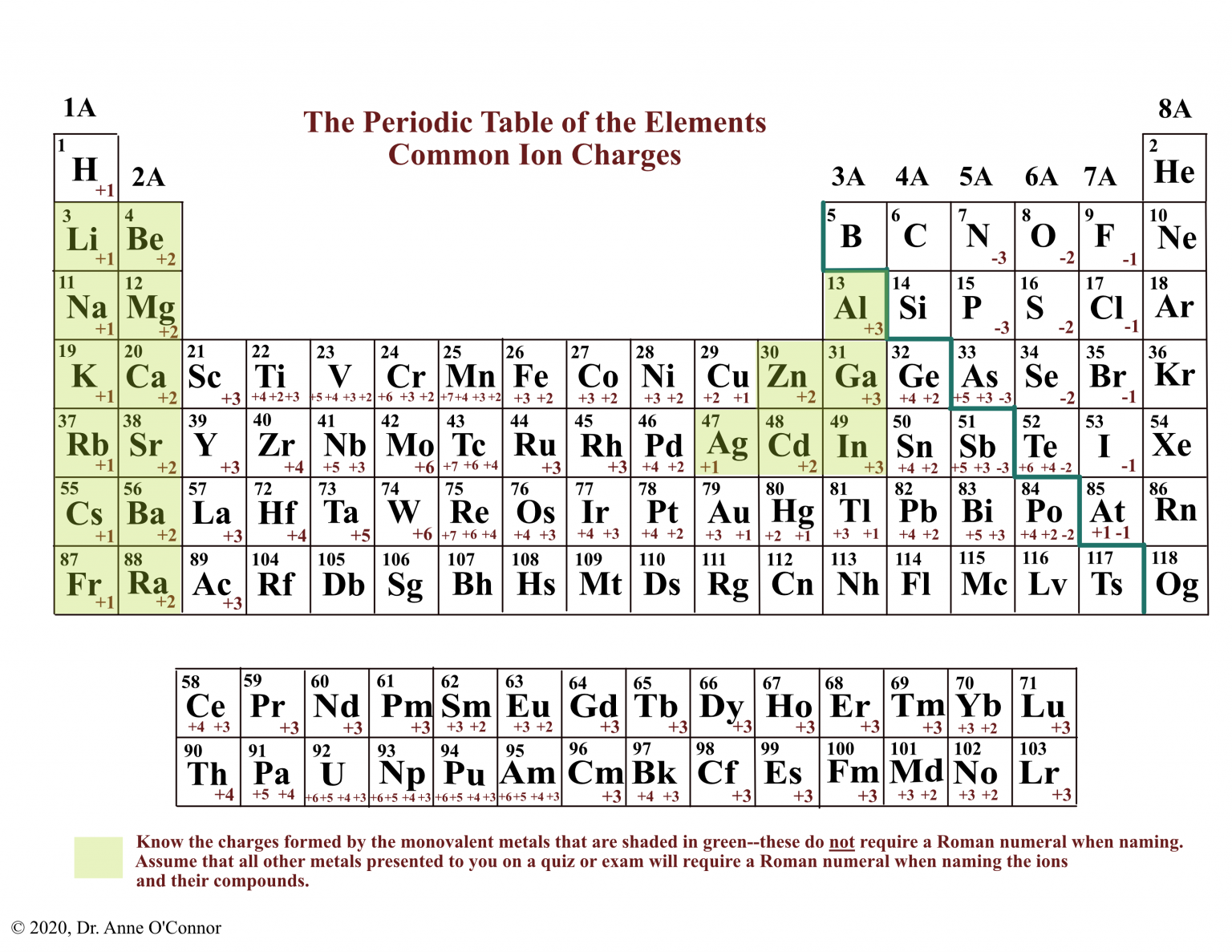
In contrast, group 17a, which consists of halogens, always forms. Aluminum, a member of the iiia family, loses three electrons to form a 3+. Web what do metals form? Oxygen often exists in a neutral state, but oxygen atoms tend to form. Web cations are positively charged ions. Web alkaline metals on earth (the iia elements) lose two electrons to form a 2+ cation. These ions are negative because they contain more electrons than protons. In these compounds you have the cation, often an alkali metal itself, stabilized in a. Elements on the left side of the periodic table tend to make cations, compared to the right side of the periodic. All metals will form cations.
Loses 1 electron to form 1+ ion. Aluminum, a member of the iiia family, loses three electrons to form a 3+. Web atoms gain electrons in their outer shell when they form negative ions, called anions. Web group 1a and 2a of the periodic table, alkali metals and alkaline earth metals respectively, always form cations. Web what do metals form? In these compounds you have the cation, often an alkali metal itself, stabilized in a. Nonmetals will form anions, except for the noble gases,. In contrast, group 17a, which consists of halogens, always forms. Elements on the left side of the periodic table tend to make cations, compared to the right side of the periodic. These ions are negative because they contain more electrons than protons.
17 Best images about What is an ion? on Pinterest Models, Shape and
Loses 1 electron to form 1+ ion. Web tara_donoghue terms in this set (60) what distinguishes a neutral atom from an ion ion has a different number of electrons do nonmetals form anions or cations? Web most transition metals differ from the metals of groups 1, 2, and 13 in that they are capable of forming more than one cation.
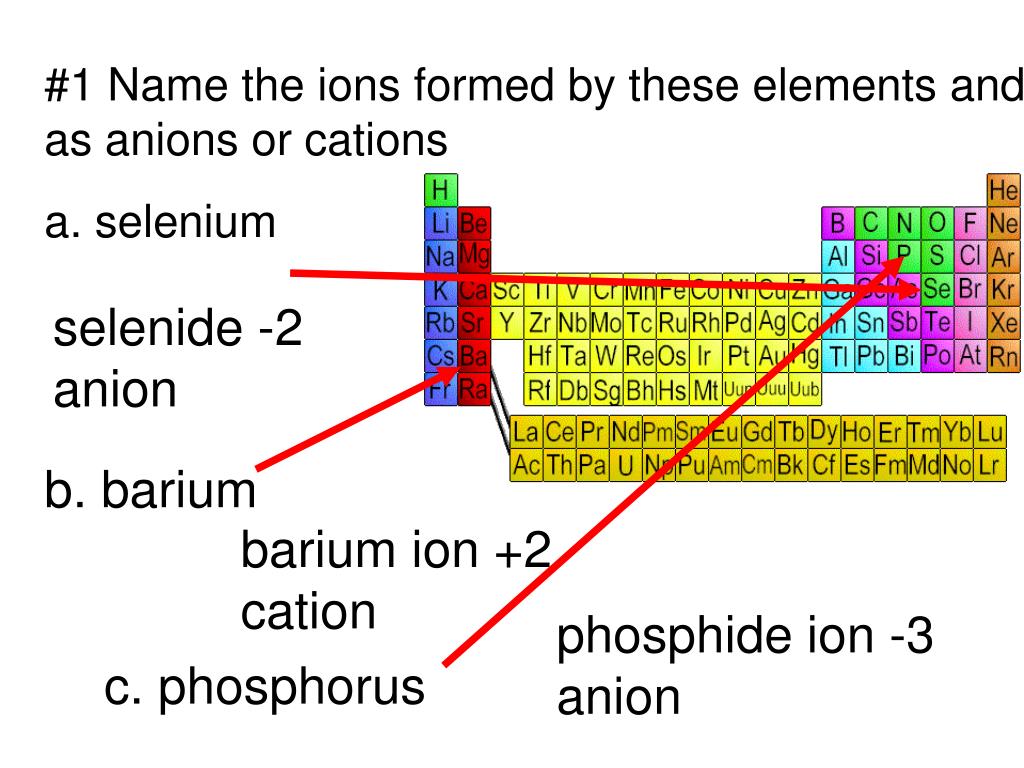
PPT 1 Name the ions formed by these elements and classify them as
Aluminum, a member of the iiia family, loses three electrons to form a 3+. Web what do metals form? Web group 1a and 2a of the periodic table, alkali metals and alkaline earth metals respectively, always form cations. Elements on the left side of the periodic table tend to make cations, compared to the right side of the periodic. Web.
anion Common anions, their names, formulas and the elements they are
Web a cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. In contrast, group 17a, which consists of halogens, always forms. Web alkalis and alkali earth metals always tend to form cations whereas halogens always tend to form anions. Web most transition metals differ from.
2.6 Ionic Compounds and Formulas Chemistry LibreTexts
Web atoms gain electrons in their outer shell when they form negative ions, called anions. Web what do metals form? All metals will form cations. Aluminum, a member of the iiia family, loses three electrons to form a 3+. Web tara_donoghue terms in this set (60) what distinguishes a neutral atom from an ion ion has a different number of.
Do metals form anions or cations quizlet? Book Vea
Anions are negatively charged ions. Oxygen often exists in a neutral state, but oxygen atoms tend to form. Web group 1a and 2a of the periodic table, alkali metals and alkaline earth metals respectively, always form cations. Web many elements can take the form of either anions or cations depending on the situation. Web alkaline metals on earth (the iia.
Form a table of alkaline earth metal cations and the
Aluminum, a member of the iiia family, loses three electrons to form a 3+. Loses 1 electron to form 1+ ion. Anions are negatively charged ions. Web cations are positively charged ions. Web most transition metals differ from the metals of groups 1, 2, and 13 in that they are capable of forming more than one cation with different ionic.
PPT Chapter 9 The Basics of Chemical Bonding PowerPoint Presentation
Web atoms gain electrons in their outer shell when they form negative ions, called anions. In contrast, group 17a, which consists of halogens, always forms. Web cations are positively charged ions. Web group 1a and 2a of the periodic table, alkali metals and alkaline earth metals respectively, always form cations. In these compounds you have the cation, often an alkali.
Nonmetals and anion formation YouTube
Loses 2 electrons to form 2+ ions. Web many elements can take the form of either anions or cations depending on the situation. Web most transition metals differ from the metals of groups 1, 2, and 13 in that they are capable of forming more than one cation with different ionic charges. Web atoms gain electrons in their outer shell.
Naming Simple Ionic Compounds Pathways to Chemistry
Web group 1a and 2a of the periodic table, alkali metals and alkaline earth metals respectively, always form cations. Web many elements can take the form of either anions or cations depending on the situation. In contrast, group 17a, which consists of halogens, always forms. Web alkaline metals on earth (the iia elements) lose two electrons to form a 2+.
PPT 1 Name the ions formed by these elements and classify them as
Web alkalis and alkali earth metals always tend to form cations whereas halogens always tend to form anions. Web cations are positively charged ions. Web group 1a and 2a of the periodic table, alkali metals and alkaline earth metals respectively, always form cations. Web many elements can take the form of either anions or cations depending on the situation. Loses.
Anions Are Negatively Charged Ions.
In contrast, group 17a, which consists of halogens, always forms. Web atoms gain electrons in their outer shell when they form negative ions, called anions. Web many elements can take the form of either anions or cations depending on the situation. Loses 1 electron to form 1+ ion.
Aluminum, A Member Of The Iiia Family, Loses Three Electrons To Form A 3+.
Oxygen often exists in a neutral state, but oxygen atoms tend to form. Loses 2 electrons to form 2+ ions. Web tara_donoghue terms in this set (60) what distinguishes a neutral atom from an ion ion has a different number of electrons do nonmetals form anions or cations? Web what do metals form?
Nonmetals Will Form Anions, Except For The Noble Gases,.
Web alkaline metals on earth (the iia elements) lose two electrons to form a 2+ cation. Web cations are positively charged ions. Web group 1a and 2a of the periodic table, alkali metals and alkaline earth metals respectively, always form cations. These ions are negative because they contain more electrons than protons.
Elements On The Left Side Of The Periodic Table Tend To Make Cations, Compared To The Right Side Of The Periodic.
Web a cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Web alkalis and alkali earth metals always tend to form cations whereas halogens always tend to form anions. Web most transition metals differ from the metals of groups 1, 2, and 13 in that they are capable of forming more than one cation with different ionic charges. In these compounds you have the cation, often an alkali metal itself, stabilized in a.