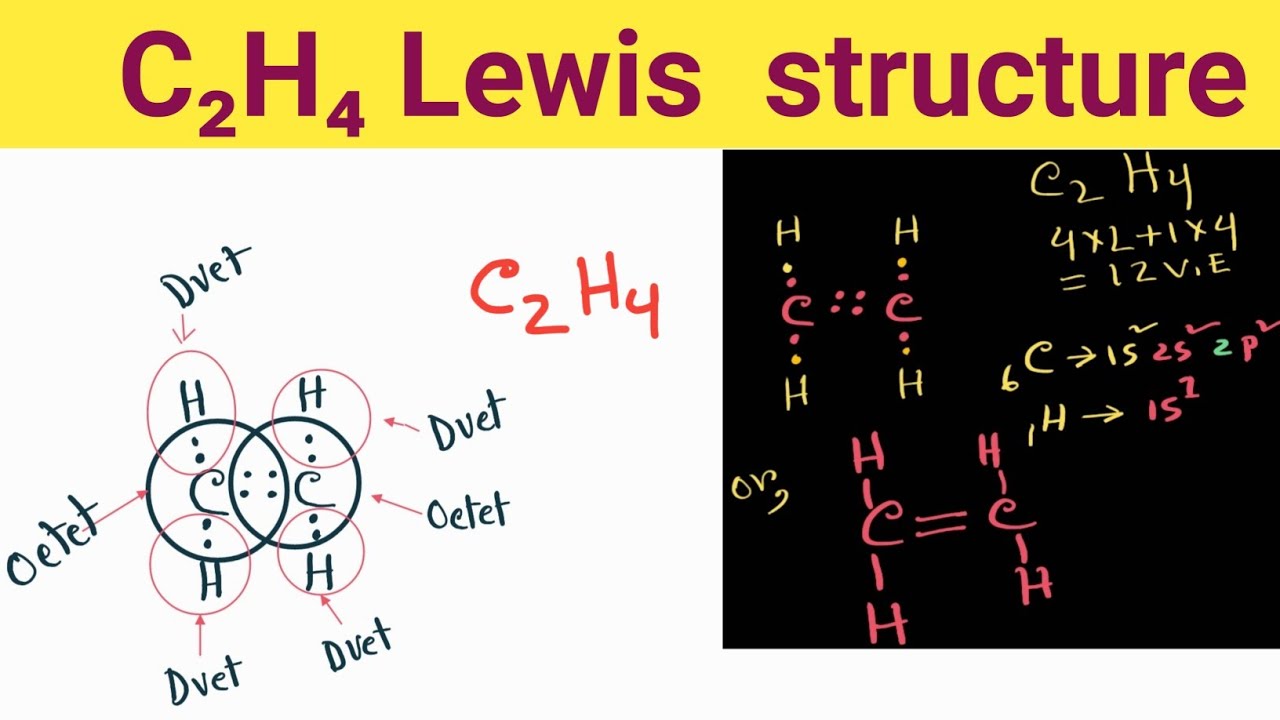
Draw The Lewis Structure Of C2H4
Draw The Lewis Structure Of C2H4 - C2h4 (one double bond), c4h6 (two double bonds), and c4h6 (one triple bond). Web draw a skeleton structure of the molecule or ion, arranging the atoms around a central atom. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web draw lewis structures for the following molecular formulas: #1 first draw a rough sketch. Draw one structure per sketcher box. This molecule is also represented by h2c=ch2, clearly showing the alkene nature of the compound. Most stable structure is taken as the lewis structure of ethene. Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Hybridization of atoms in ethene molecue can be found from lewis structure.
This widget gets the lewis structure of chemical compounds. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Having a double c=c bond, it is unsaturated and this gives rise to several properties. Here, we learned about how to draw the proper lewis structure and find out the molecular geometry of an ethylene molecule. There are 2 steps to solve this one. Note that the c 2 h 4 lewis dot structure involves sharing more than one pair of electrons. Draw lewis structures for the ethylene molecule (c2h4), the chloroethyne molecule (c2hcl), and the acetaldehyde molecule (ch3cho) in the window below, and then answer the questions that follow, based on your drawings. Web for very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the constituent atoms. (please note, none of the solutions are using the expanded octet rule or formal charges) h 2. How many lone pairs are on the central atom in the perchlorate ion?
Web use these steps to correctly draw the c 2 h 4 lewis structure: Web how to draw the lewis dot structure for c2h4: Let’s draw and understand this lewis dot structure step by step. First, we need to draw the lewis structure of c 2 h 4. #3 calculate and mark formal charges on the atoms, if required. Find more chemistry widgets in wolfram|alpha. #5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure. Web for very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the constituent atoms. Here, we learned about how to draw the proper lewis structure and find out the molecular geometry of an ethylene molecule. How many lone pairs are on the central atom in the perchlorate ion?
draw lewis structures for the ethylene molecule ( c2h4 ), the
Find more chemistry widgets in wolfram|alpha. Hybridization of atoms in ethene molecue can be found from lewis structure. There are 2 steps to solve this one. #2 mark lone pairs on the atoms. Each step of determining the lewis structure of ethene and hybridization are explained in this tutorial.
C2H4 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram
Draw lewis structures for the ethylene molecule (c2h4), the chloroethyne molecule (c2hcl), and the acetaldehyde molecule (ch3cho) in the window below, and then answer the questions that follow, based on your drawings. For the c2h4 structure use the periodic table to find the total number of valence electrons for the c2h4. This widget gets the lewis structure of chemical compounds..
C2H4 Lewis StructureHow do you draw the Lewis structure for C2H4
Web how to draw the lewis dot structure for c2h4: There are 2 steps to solve this one. Web draw the lewis structures of c2h6, c2h4, and c2h2. Here, we learned about how to draw the proper lewis structure and find out the molecular geometry of an ethylene molecule. Web this video teaches you how to draw the lewis structures.
C2H4 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram
#2 mark lone pairs on the atoms. Web ethene's lewis structure can be built by vsepr rule. The chemical formula c2h4 represents ethylene. Steps of drawing c2h4 lewis structure. (please note, none of the solutions are using the expanded octet rule or formal charges) h 2.
C2H4 Lewis Structure (Ethylene) YouTube
Most stable structure is taken as the lewis structure of ethene. #1 first draw a rough sketch. Web draw lewis structures for the following molecular formulas: Web draw a skeleton structure of the molecule or ion, arranging the atoms around a central atom. (generally, the least electronegative element should be placed in the center.) connect each atom to the central.
C2H4 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram
Web for very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the constituent atoms. First, we need to draw the lewis structure of c 2 h 4. C2h4 exists as a colorless gas and is. What is the lewis dot symbol for lead? For the c2h4 structure use the.
Hướng dẫn cách vẽ c2h4 lewis structure đúng và đầy đủ
Web for very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the constituent atoms. #2 mark lone pairs on the atoms. Web write lewis structures for the following: Draw one structure per sketcher box. Draw lewis structures for the ethylene molecule (c2h4), the chloroethyne molecule (c2hcl), and the acetaldehyde.
Is C2H4 Polar or Nonpolar? YouTube
Web learn the steps to draw the lewis structure of c2h4 (ethene) in just 1 minute.📌you can draw any lewis structures by following the simple steps mentioned in. In order to find the total valence electrons in c2h4 molecule, first of all you should know the valence electrons present in carbon atom as well as hydrogen atom. Web use these.
C2h4 Dot Diagram
Web learn the steps to draw the lewis structure of c2h4 (ethene) in just 1 minute.📌you can draw any lewis structures by following the simple steps mentioned in. Web watch the video of dr. Steps of drawing c2h4 lewis structure. Web c2h4 geometry and hybridization. C2h4 (one double bond), c4h6 (two double bonds), and c4h6 (one triple bond).
Draw the Lewis structure for C2H4 (whose skeletal structure is H2CCH2
C2h4 exists as a colorless gas and is. Web write lewis structures for the following: Find the total valence electrons in c2h4 molecule. #5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure. (generally, the least electronegative element should be placed in the center.) connect each atom to the central atom with a.
Find More Chemistry Widgets In Wolfram|Alpha.
There are 2 steps to solve this one. Most stable structure is taken as the lewis structure of ethene. Each step of determining the lewis structure of ethene and hybridization are explained in this tutorial. Web drawing lewis structures for molecules with one central atom:
Steps Of Drawing C2H4 Lewis Structure.
Web draw lewis structures for the following molecular formulas: #2 mark lone pairs on the atoms. Find the total valence electrons in c2h4 molecule. Draw lewis structures for the ethylene molecule (c2h4), the chloroethyne molecule (c2hcl), and the acetaldehyde molecule (ch3cho) in the window below, and then answer the questions that follow, based on your drawings.
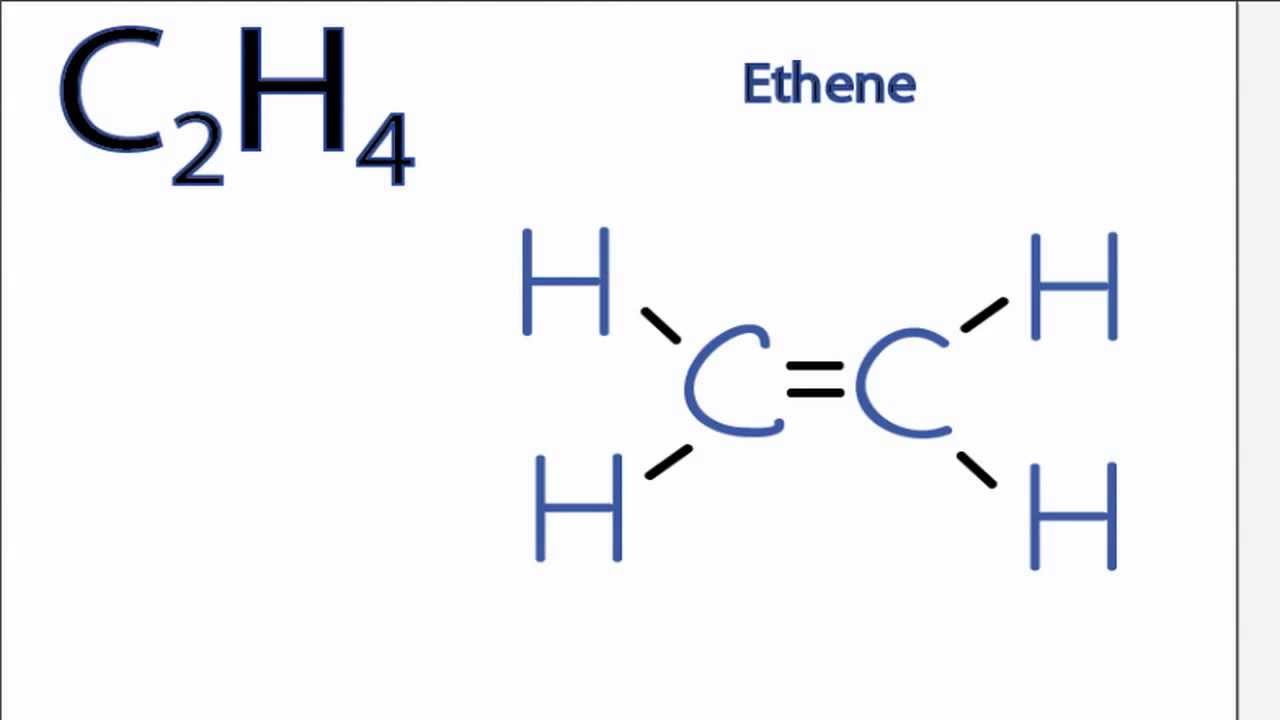
Web Lewis Structure Of C2H4 (Or Ethene) Contains One Double Bond Between The Two Carbon (C) Atoms And Four Single Bonds Between Carbon (C) & Hydrogen (H) Atoms.
Note that the c 2 h 4 lewis dot structure involves sharing more than one pair of electrons. Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Web how would you draw a lewis structure for an atom that has the electron configuration #1s^2 2s^2 2p^6 3s^2 3p^3#? #3 calculate and mark formal charges on the atoms, if required.
Here, We Learned About How To Draw The Proper Lewis Structure And Find Out The Molecular Geometry Of An Ethylene Molecule.
#5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure. What is the lewis dot symbol for lead? In order to find the total valence electrons in c2h4 molecule, first of all you should know the valence electrons present in carbon atom as well as hydrogen atom. Web write lewis structures for the following: