Electron Drawings
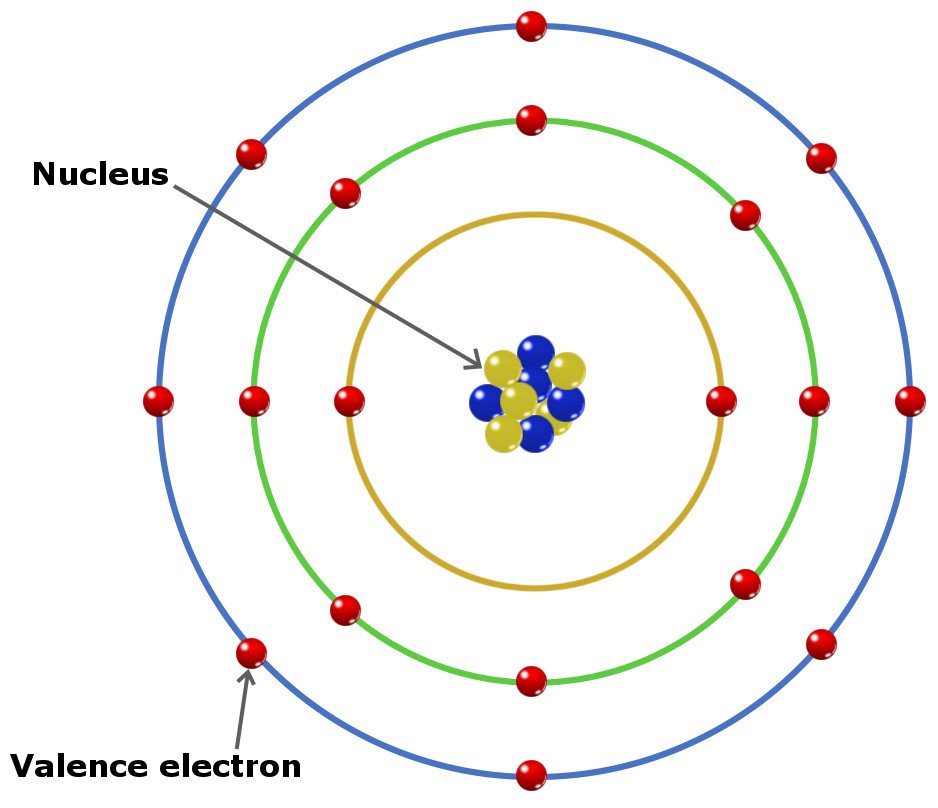
Electron Drawings - A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. In polar reaction mechanisms, such as the nucleophilic substitution reactions of haloalkanes, electron flow will be designated by arrows indicating the movement of electrons from electron rich regions to electron poor regions. Electrons are represented by dots or crosses and are positioned in energy levels, or ‘shells’, around the central nucleus. Web here are electron shell atom diagrams for the elements, ordered by increasing atomic number. Web an electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Web electron configuration diagrams | properties of matter | chemistry | fuseschoollearn the basics about drawing electron configuration diagrams. Then, compare the model to real molecules! Find out by adding single, double or triple bonds and lone pairs to the central atom. This diagram shows the valence electrons as dots around the symbol. Web electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the element’s symbol.
Then, compare the model to real molecules! Web we construct the periodic table by following the aufbau principle (from german, meaning “building up”). Find out by adding single, double or triple bonds and lone pairs to the central atom. Web a lewis electron dot diagram (or electron dot diagram or a lewis diagram or a lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The alkali metal sodium (atomic number 11) has one more electron than the neon atom. Web an electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Orbital diagrams are a visual way to show where the electrons are located within an atom. When reading the periodic table from left to right, one can easily write an electron. Web draw a lewis electron dot symbol for a given atom. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule.
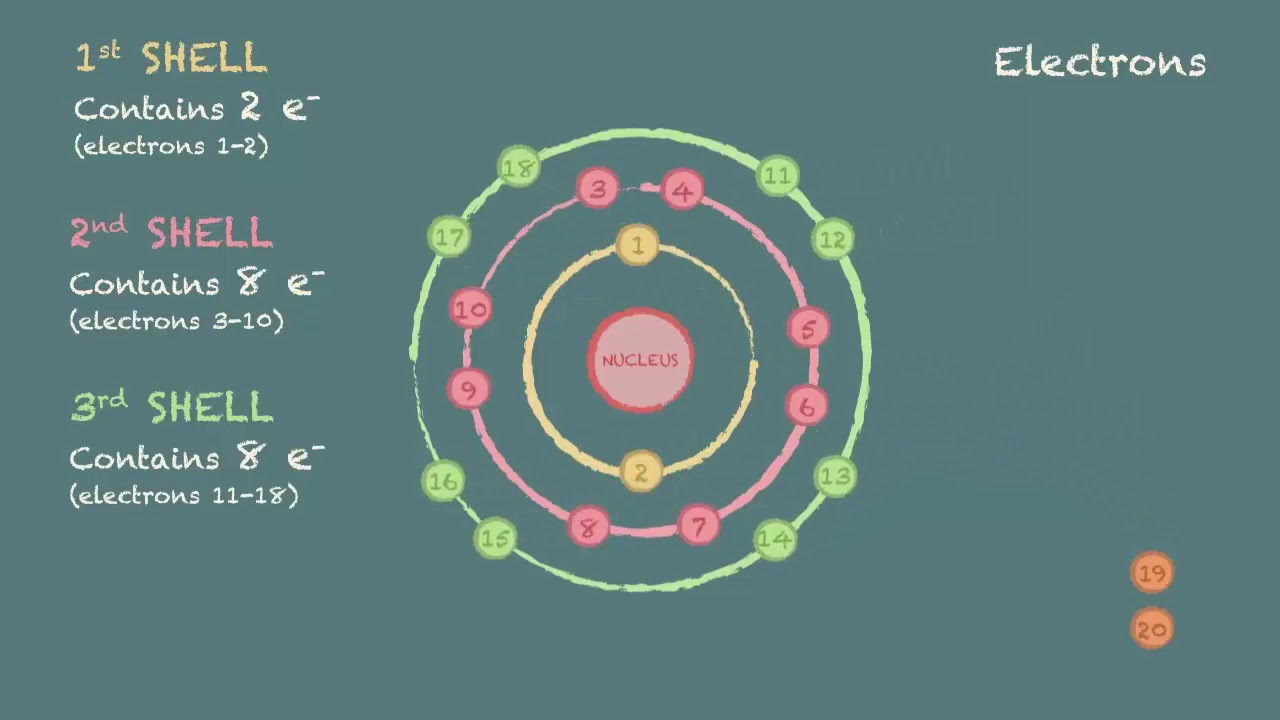
When reading the periodic table from left to right, one can easily write an electron. The number of dots equals. An electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Web an electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Electrons are represented by dots or crosses and are positioned in energy levels, or ‘shells’, around the central nucleus. Web a simple tutorial on how to draw electron configurations efficiently, and how they are written. Web we construct the periodic table by following the aufbau principle (from german, meaning “building up”). Web how to draw and write electronic structures (also called the electron configurations).an essential skill if you are studying gcse chemistry or gcse combined. 366k views 10 years ago chemistry tutorials: Web electron configurations and orbital box diagrams can be written right from the periodic table.
What Are Valence Electrons And How To Find Them? Where Are They Located?
Typically, they only show the outermost electrons. For each electron shell atom diagram, the element symbol is listed in the nucleus. Web a simple tutorial on how to draw electron configurations efficiently, and how they are written. Then, compare the model to real molecules! The number of dots equals the number of valence electrons in the atom.
Lets Get Inside An Atom!! The Science Station
Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. An electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Web electron configurations and orbital box diagrams can be written right from the periodic table. Web in.
Drawing electron configuration diagrams YouTube
Then, compare the model to real molecules! Typically, they only show the outermost electrons. A beryllium atom, with two valence electrons, would have the electron dot diagram below. Find out by adding single, double or triple bonds and lone pairs to the central atom. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence.
Electronic structure of matter. San Francisco de Paula, Science
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. Electrons are represented by dots or crosses and are positioned in energy levels, or ‘shells’, around the central nucleus. Web draw a lewis electron dot symbol for a given atom. Web a lewis electron dot diagram (or electron dot diagram or a lewis diagram or.
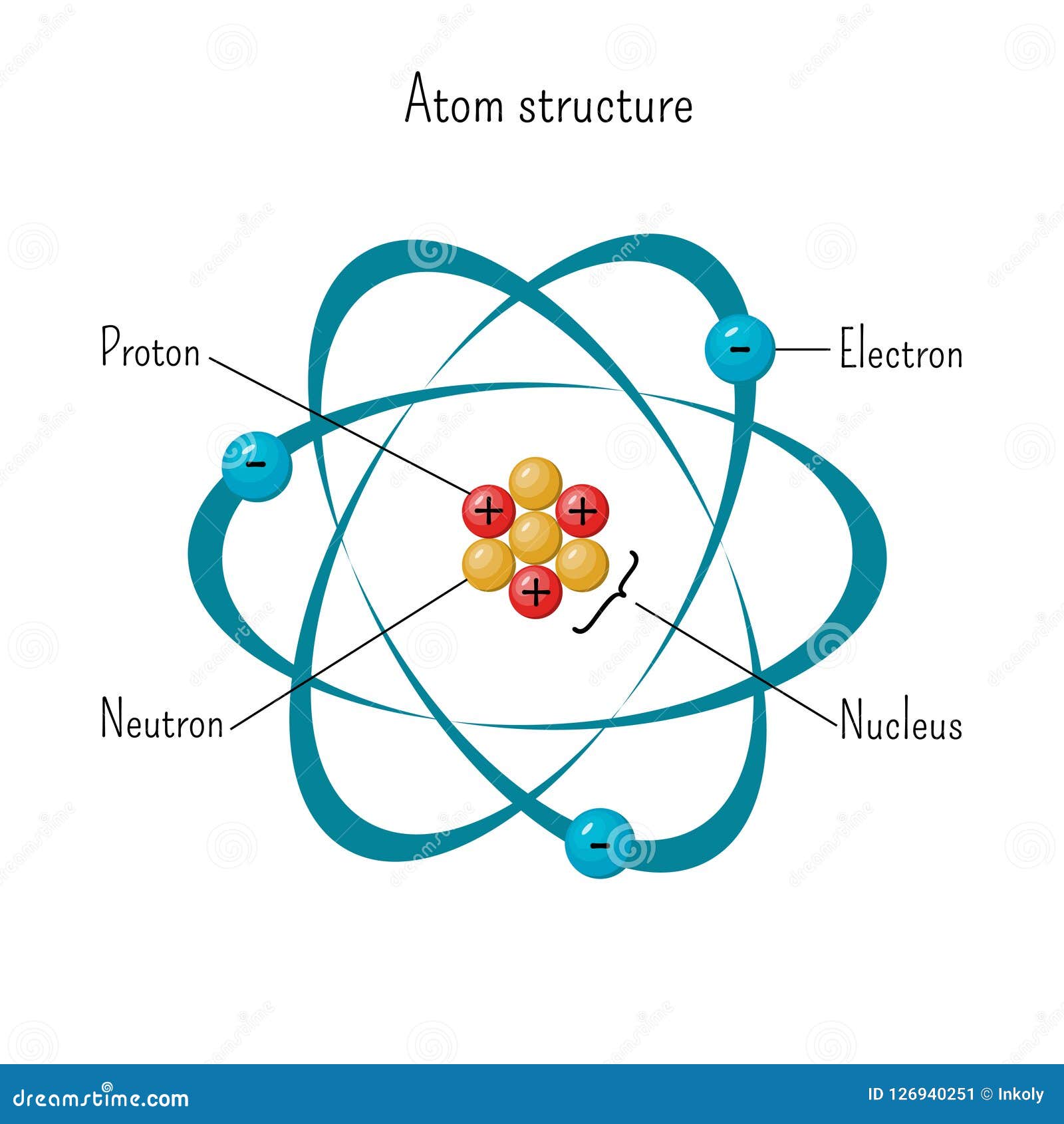
Simple Model of Atom Structure with Electrons Orbiting Nucleus of Three
Web electron configurations and orbital box diagrams can be written right from the periodic table. In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. How does molecule shape change with different numbers of bonds and electron pairs? When reading the periodic table from left to right, one can easily write an electron. Web electron.
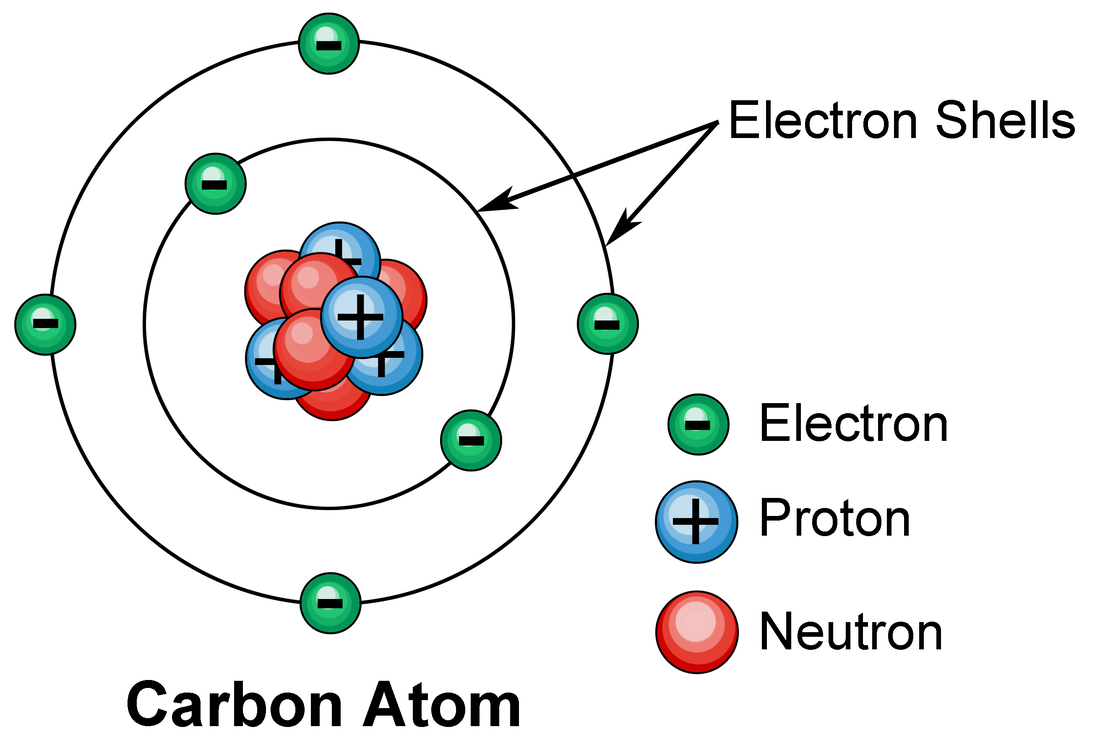
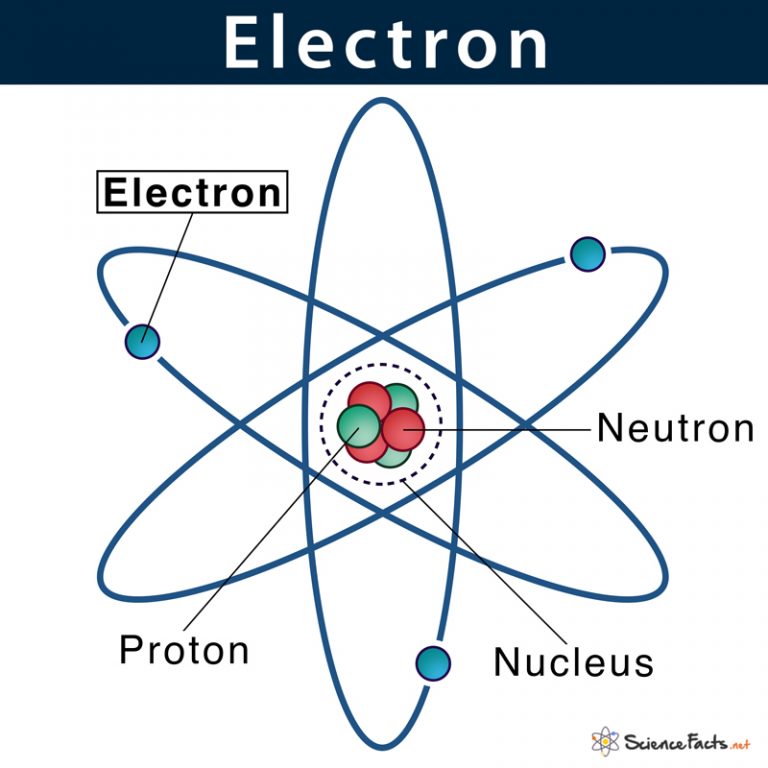
Electrons Biology for Majors I
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. This diagram shows the valence electrons as dots around the symbol. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. 366k views 10 years ago chemistry tutorials: Web electron configurations and orbital.
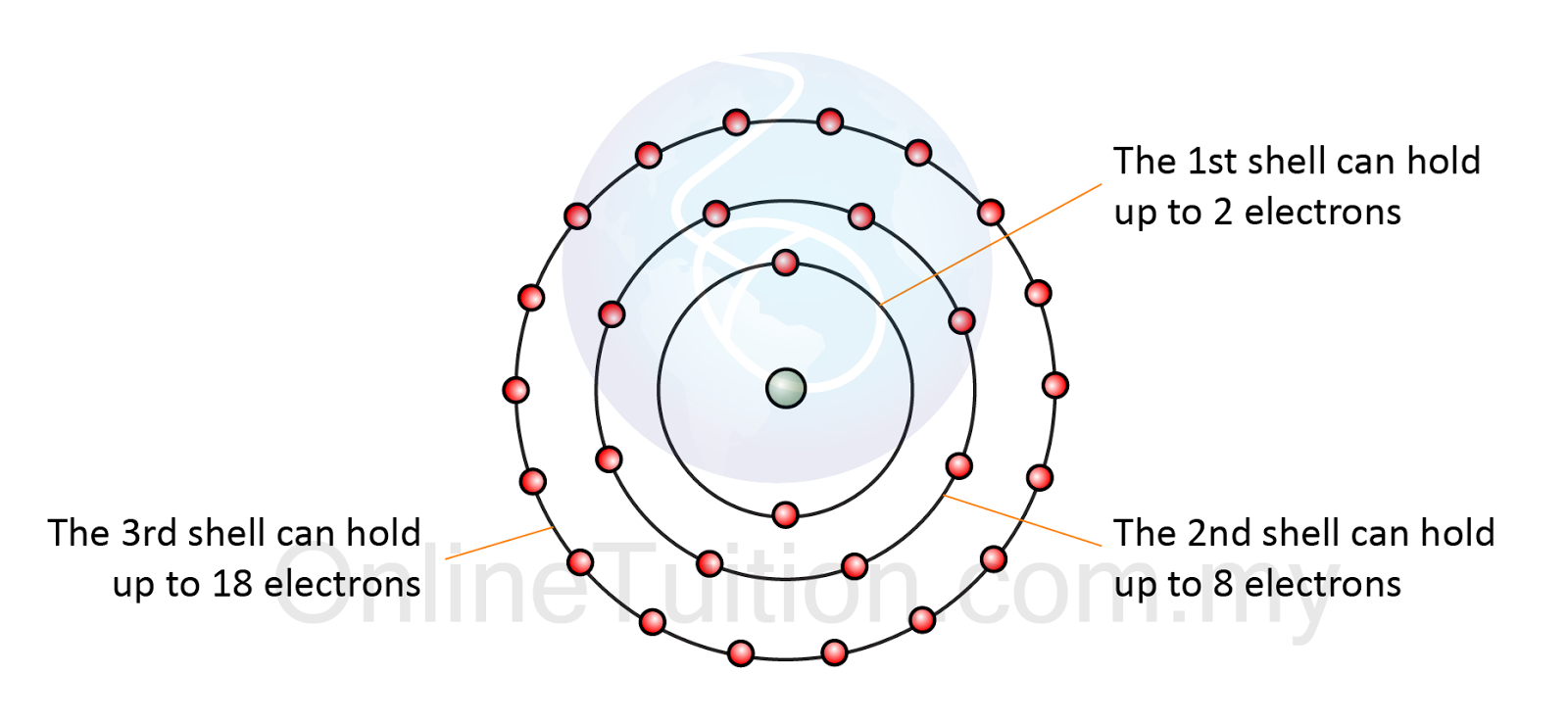
Electron Arrangement in Atom SPM Chemistry
Typically, they only show the outermost electrons. Web this chemistry tutorial video shows you how to draw an electron dot diagram or a lewis dot diagram for an atom. Web the easiest way to create electron configurations is using an electron configuration table, which is a way of writing down the various orbitals available to electrons. Web a simple tutorial.
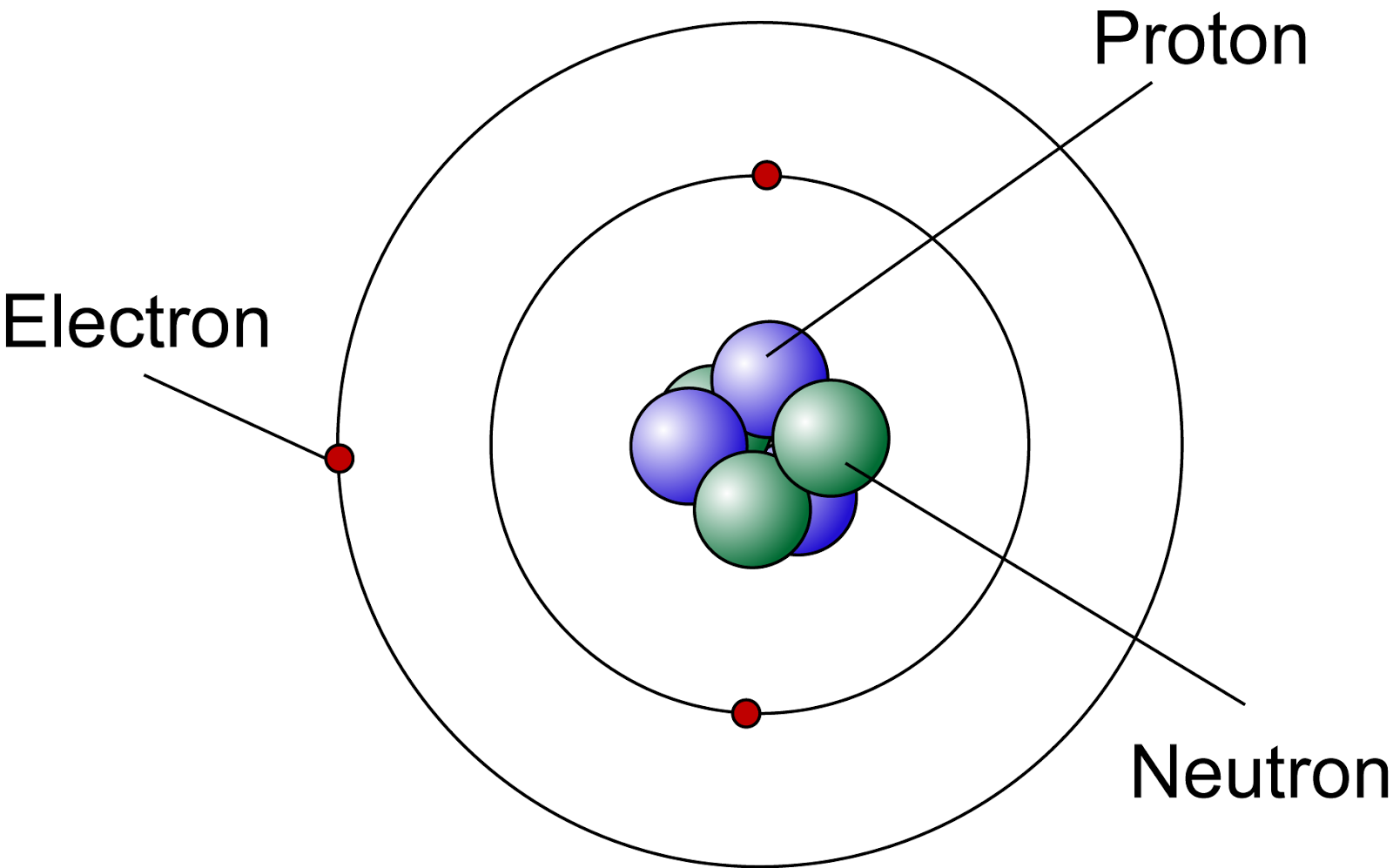
Atomic Nucleus Definition, Structure & Parts with Diagram
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. The alkali metal sodium (atomic number 11) has one more electron than the neon atom. Web electron configuration diagrams | properties of matter | chemistry | fuseschoollearn the basics about drawing electron configuration diagrams. Electrons are represented by dots or crosses and are positioned in.
Electron Arrangement in Atom SPM Chemistry
Web an electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. In polar reaction mechanisms, such as the nucleophilic substitution reactions of haloalkanes, electron flow will be designated by arrows indicating the movement of electrons from electron rich regions to electron poor regions. Electrons are represented by dots or.
Atom Diagrams Electron Configurations of the Elements
When reading the periodic table from left to right, one can easily write an electron. A beryllium atom, with two valence electrons, would have the electron dot diagram below. Web electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the element's symbol. Electrons are represented by dots or crosses and.
In Polar Reaction Mechanisms, Such As The Nucleophilic Substitution Reactions Of Haloalkanes, Electron Flow Will Be Designated By Arrows Indicating The Movement Of Electrons From Electron Rich Regions To Electron Poor Regions.
Web an electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Web electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the element’s symbol. Find out by adding single, double or triple bonds and lone pairs to the central atom. Web a lewis electron dot diagram (or electron dot diagram, or a lewis diagram, or a lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element.
The Number Of Dots Equals.
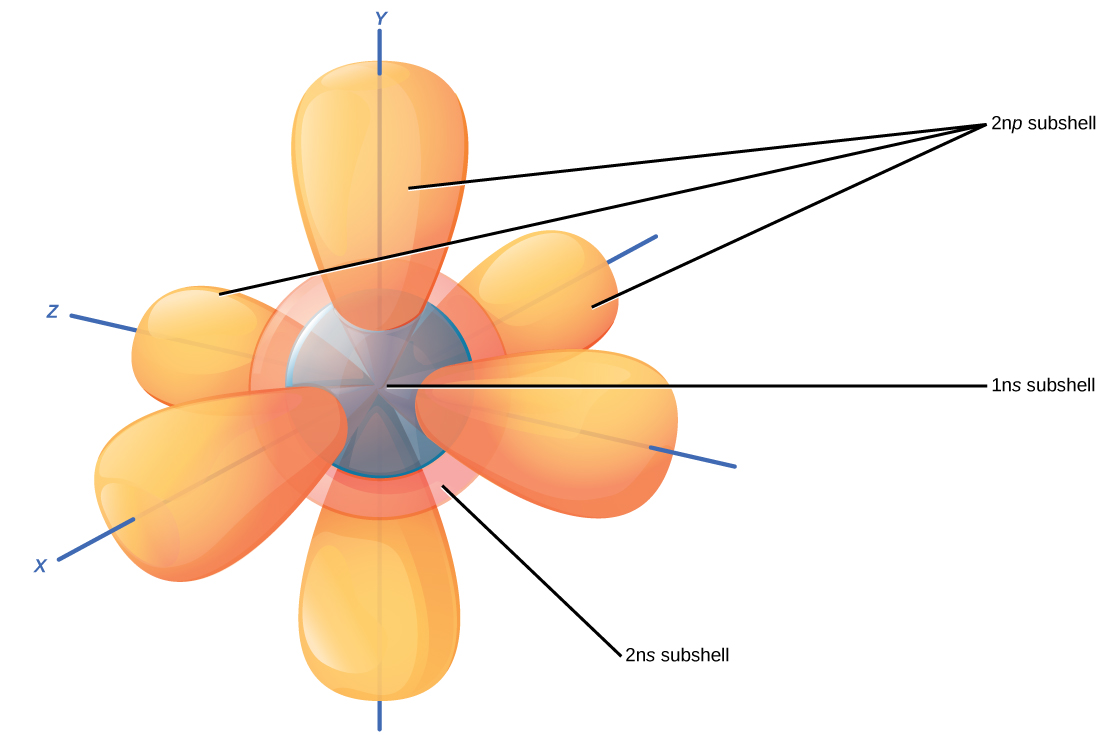
For example, the electron configuration of lithium, 1 s ²2 s ¹, tells us that lithium has two electrons in the 1 s subshell and one electron in the 2 s subshell. This article will explore the basics of how to draw each type of diagram, and important rules to. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. Web here are electron shell atom diagrams for the elements, ordered by increasing atomic number.
Then, Compare The Model To Real Molecules!
A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Electrons are represented by dots or crosses and are positioned in energy levels, or ‘shells’, around the central nucleus. An electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Web we construct the periodic table by following the aufbau principle (from german, meaning “building up”).
Web The Easiest Way To Create Electron Configurations Is Using An Electron Configuration Table, Which Is A Way Of Writing Down The Various Orbitals Available To Electrons.
Electrons are represented by dots or crosses and are positioned in energy levels, or ‘shells’, around the central nucleus. Web draw a lewis electron dot symbol for a given atom. Web this chemistry tutorial video shows you how to draw an electron dot diagram or a lewis dot diagram for an atom. Orbital diagrams must follow 3 rules:

:max_bytes(150000):strip_icc()/chlorineatom-58b602515f9b5860464c5c02.jpg)