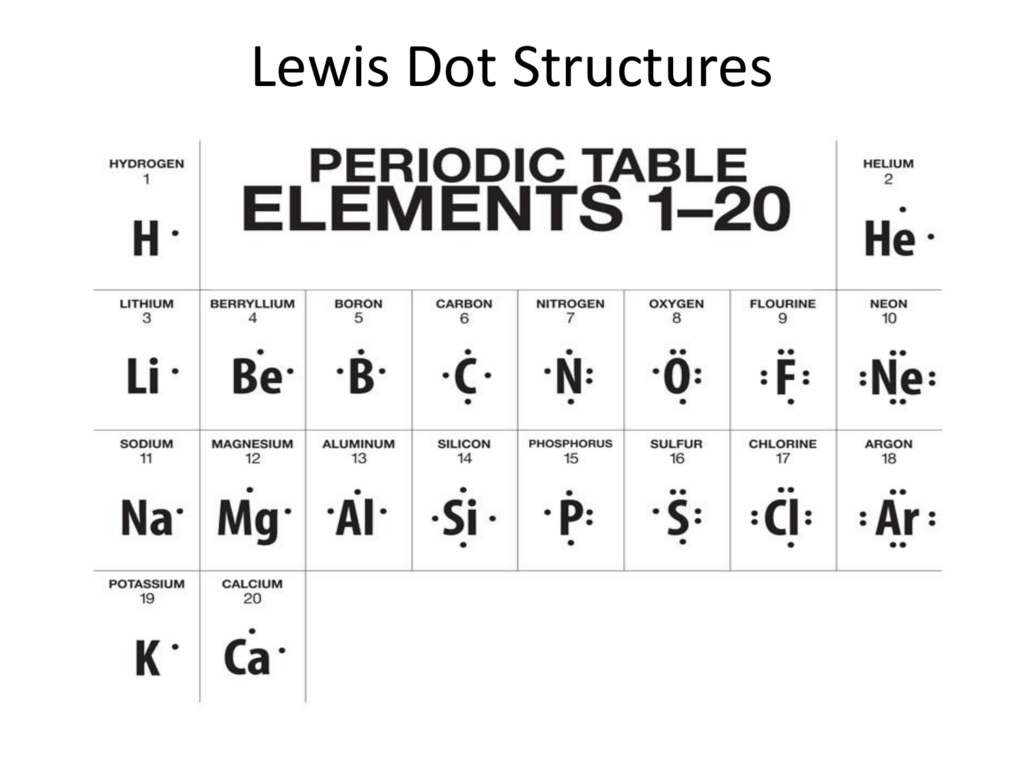
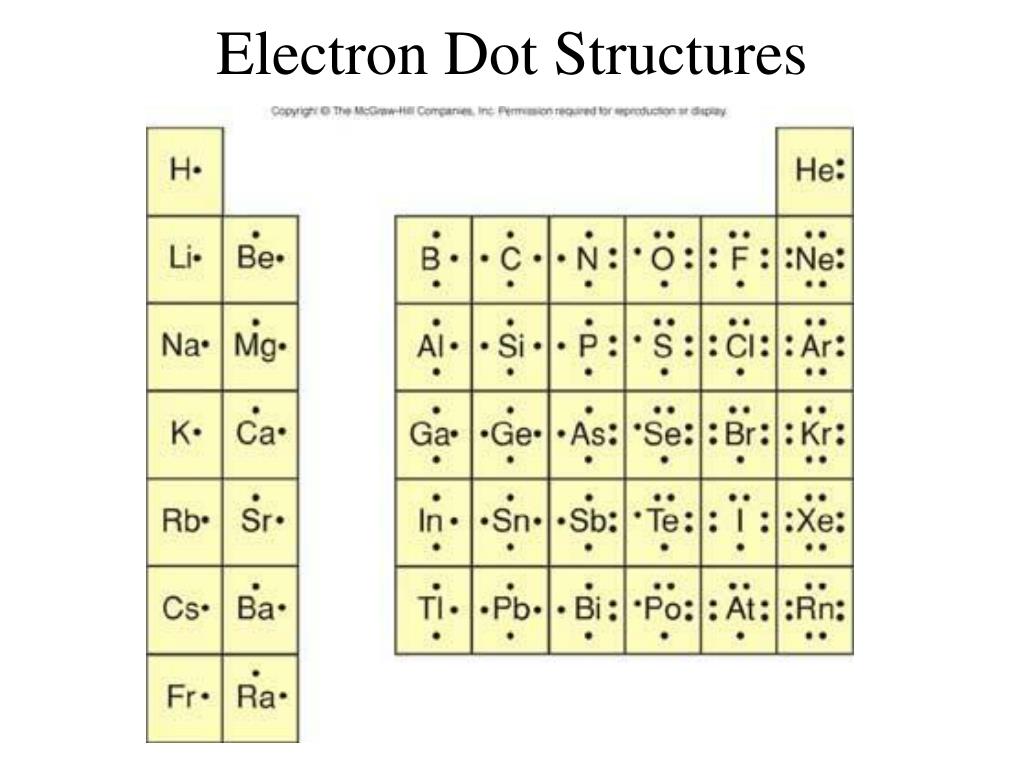
How Do You Draw An Electron Dot Diagram
How Do You Draw An Electron Dot Diagram - The number of dots equals. You can alternate the dots and crosses in simple diagrams or use other colours or symbols for larger diagrams. In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. More complicated versions can be used to show the bond between different atoms in a molecule. Draw a lewis electron dot diagram for an atom or a monatomic ion. And we would account for these valence electrons in our dot structure. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Web how to draw electron dot structures? Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. Web electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the element's symbol.
The first thing we would need to do is to find the total number of valence electrons. Lewis electron dot diagrams for ions have fewer (for cations) or more (for anions) dots than the corresponding atom. Write the symbol of the atom you are drawing the electron dot diagram for in the middle of your paper. The sum of the valence electrons is. Web to draw the lewis electron dot diagram we picture in our minds the symbol for mg in a box with all of its core electrons (i.e., 1 s2 2 s2 2 p6 ). Web a lewis electron dot diagram (or electron dot diagram or a lewis diagram or a lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Web an electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. You can alternate the dots and crosses in simple diagrams or use other colours or symbols for larger diagrams. Determine the total number of valence (outer shell) electrons.
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. Shared pairs of electrons are drawn as lines between atoms, while. Web how to draw electron dot structures? This dot and cross diagram shows the outer shells touching. A lewis electron dot formula comprises one dot for every valence electron and an element’s symbol. The first thing we would need to do is to find the total number of valence electrons. Stages to articulate the electron dot formula are stated beneath. Electrons are represented by dots or crosses and are positioned in energy levels, or ‘shells’, around the central nucleus. And we would account for these valence electrons in our dot structure. Web draw a lewis electron dot diagram for any atom or a monatomic ion with an atomic number of less than 20.
Valence Electrons Presentation Chemistry
Web lewis dot structures, or lewis structures for short, are visuals that represent the outermost shell of electrons, also known as valence electrons, and possible covalent bonds within an atom or molecule. Web these diagrams are used as a shorthand notation to show the number of valence electrons in an atom. These lewis symbols and lewis structures help visualize the.
BohrRutherford Diagrams & Lewis Dot Diagrams Eve Wongworakul
Draw a lewis electron dot diagram for an atom or a monatomic ion. 4.2k views 4 years ago chemistry (grade level, regents level) this chemistry tutorial video shows you how to draw an electron dot diagram or a lewis dot diagram for an atom. Web to draw the lewis structure of an atom, write the symbol of the atom and.
Lewis Dot Structures
Web draw the completed dot and cross diagram. Web here's some of the guidelines for drawing dot structures. Web a lewis structure is a diagram that shows the chemical bonds between atoms in a molecule and the valence electrons or lone pairs of electrons. Web these diagrams are used as a shorthand notation to show the number of valence electrons.
How to Draw Electron Dot Diagrams Sciencing
How many valence electrons does magnesium have? This dot and cross diagram shows the outer shells touching. The number of dots equals. If the species is an ion, add or subtract electrons corresponding to the charge. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful.
Lewis Dot Structure Definition, Examples, and Drawing
These lewis symbols and lewis structures help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds. Web lewis structures (also known as lewis dot structures or electron dot structures) are diagrams that represent the valence electrons of atoms within a molecule. In almost all cases, chemical bonds are formed by interactions of.
3 Ways to Draw Lewis Dot Structures wikiHow
Web lewis structures (also known as lewis dot structures or electron dot structures) are diagrams that represent the valence electrons of atoms within a molecule. Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. How many valence electrons does magnesium have? Draw a lewis.
PPT Drawing Lewis Structures A Tutorial on Writing Lewis Dot
Web these diagrams are used as a shorthand notation to show the number of valence electrons in an atom. Note down a skeletal structure displaying a realistic bonding pattern by means of only the element symbols. Draw a lewis electron dot diagram for an atom or a monatomic ion. Web to draw the lewis electron dot diagram we picture in.
How To Draw Lewis Dot Diagrams
Draw the atoms on paper and put dots around them to represent valence electrons of the atom. Then we place the valence electrons around the sides of the box with each side representing an orbital in the outermost energy level. Web a video tutorial for how to draw lewis structures in five steps. Web an electron configuration diagram is a.
How to Draw a Lewis Structure
Electrons are represented by dots or crosses and are positioned in energy levels, or ‘shells’, around the central nucleus. A beryllium atom, with two valence electrons, has the electron dot diagram below: A lewis electron dot formula comprises one dot for every valence electron and an element’s symbol. This dot and cross diagram shows the outer shells touching. Web to.
PPT Electron Dot Structures PowerPoint Presentation, free download
Web a lewis structure is a diagram that shows the chemical bonds between atoms in a molecule and the valence electrons or lone pairs of electrons. A lewis electron dot formula comprises one dot for every valence electron and an element’s symbol. Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots.
Web Electron Dot Diagrams Are Diagrams In Which The Valence Electrons Of An Atom Are Shown As Dots Distributed Around The Element’s Symbol.
Stages to articulate the electron dot formula are stated beneath. Web to draw the lewis electron dot diagram we picture in our minds the symbol for mg in a box with all of its core electrons (i.e., 1 s2 2 s2 2 p6 ). These lewis symbols and lewis structures help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds. This is sometimes called the bohr, or the ‘solar system’, model.
Web Here's Some Of The Guidelines For Drawing Dot Structures.
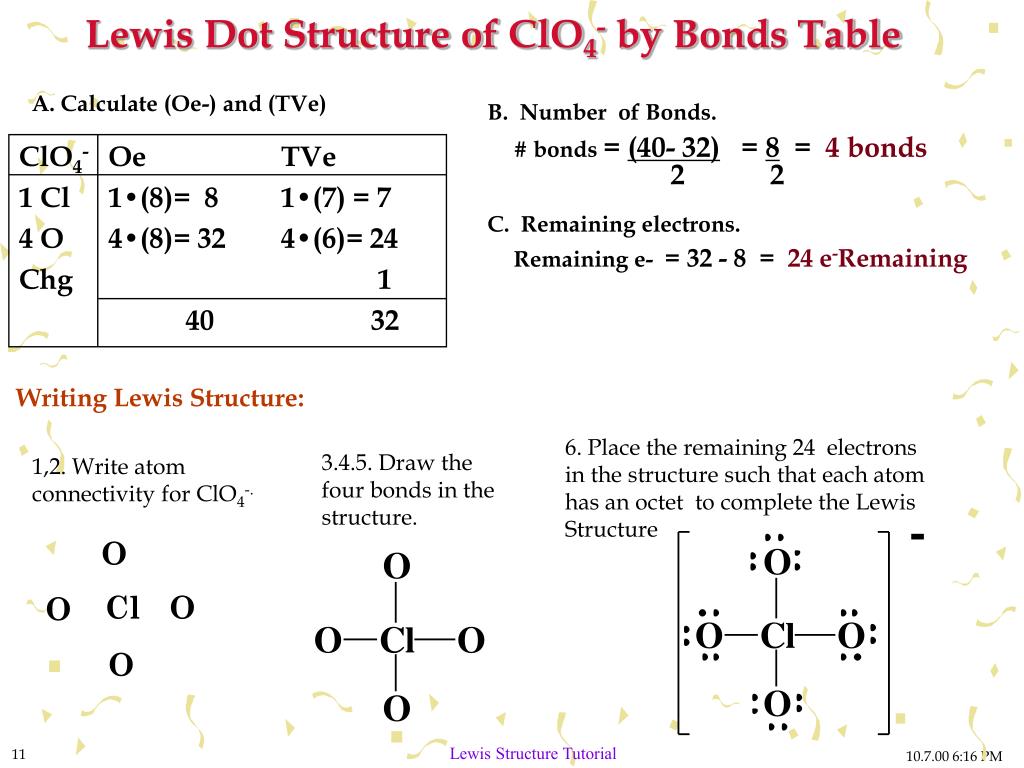
Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. More complicated versions can be used to show the bond between different atoms in a molecule. Be sure to have the correct number of electrons. So let's say we wanted to draw the dot structure for this molecule, so silicon tetrafluoride.
How Many Valence Electrons Does Magnesium Have?
You can alternate the dots and crosses in simple diagrams or use other colours or symbols for larger diagrams. Web a lewis structure is a diagram that shows the chemical bonds between atoms in a molecule and the valence electrons or lone pairs of electrons. A lewis electron dot formula comprises one dot for every valence electron and an element’s symbol. The first thing we would need to do is to find the total number of valence electrons.
Web Draw The Completed Dot And Cross Diagram.
Write the symbol of the atom you are drawing the electron dot diagram for in the middle of your paper. Web an electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. The diagram is also called a lewis dot diagram, lewis dot formula, or electron dot diagram. Shared pairs of electrons are drawn as lines between atoms, while.
.PNG)