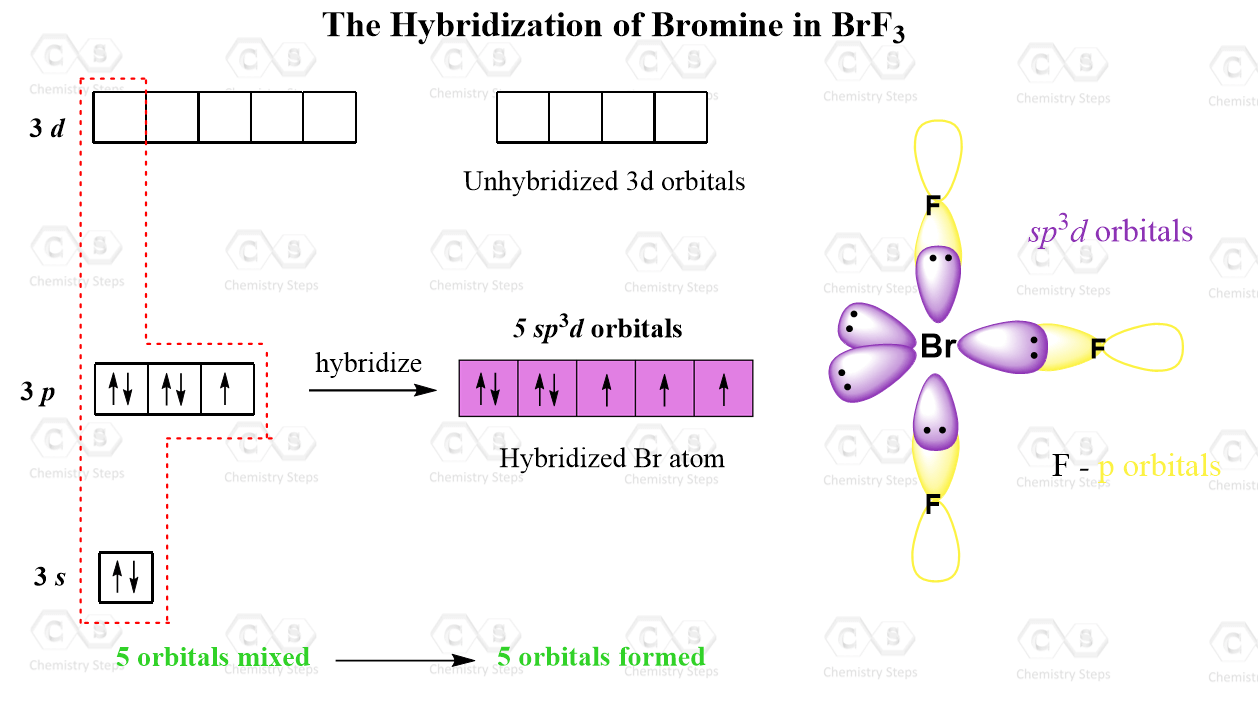
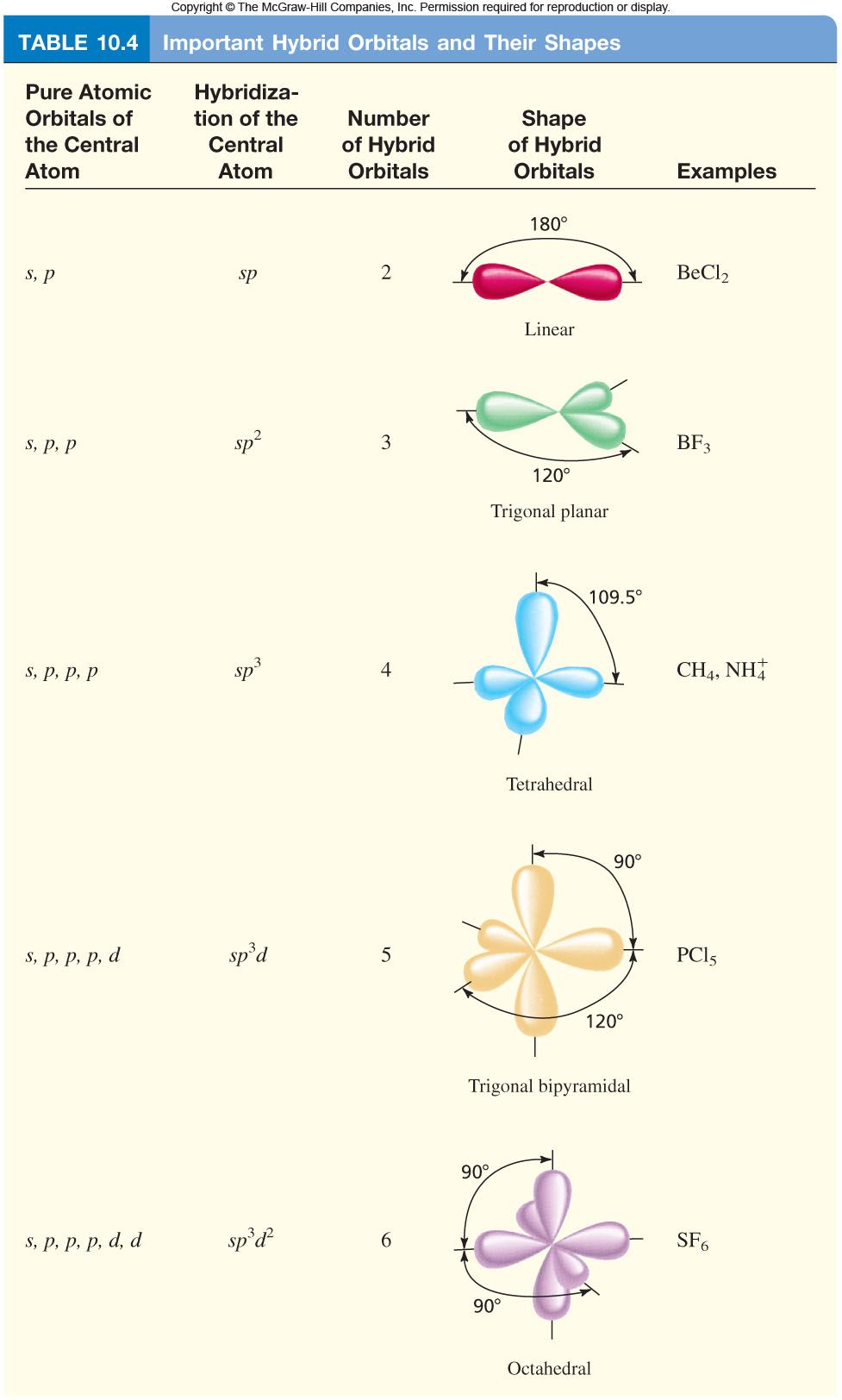
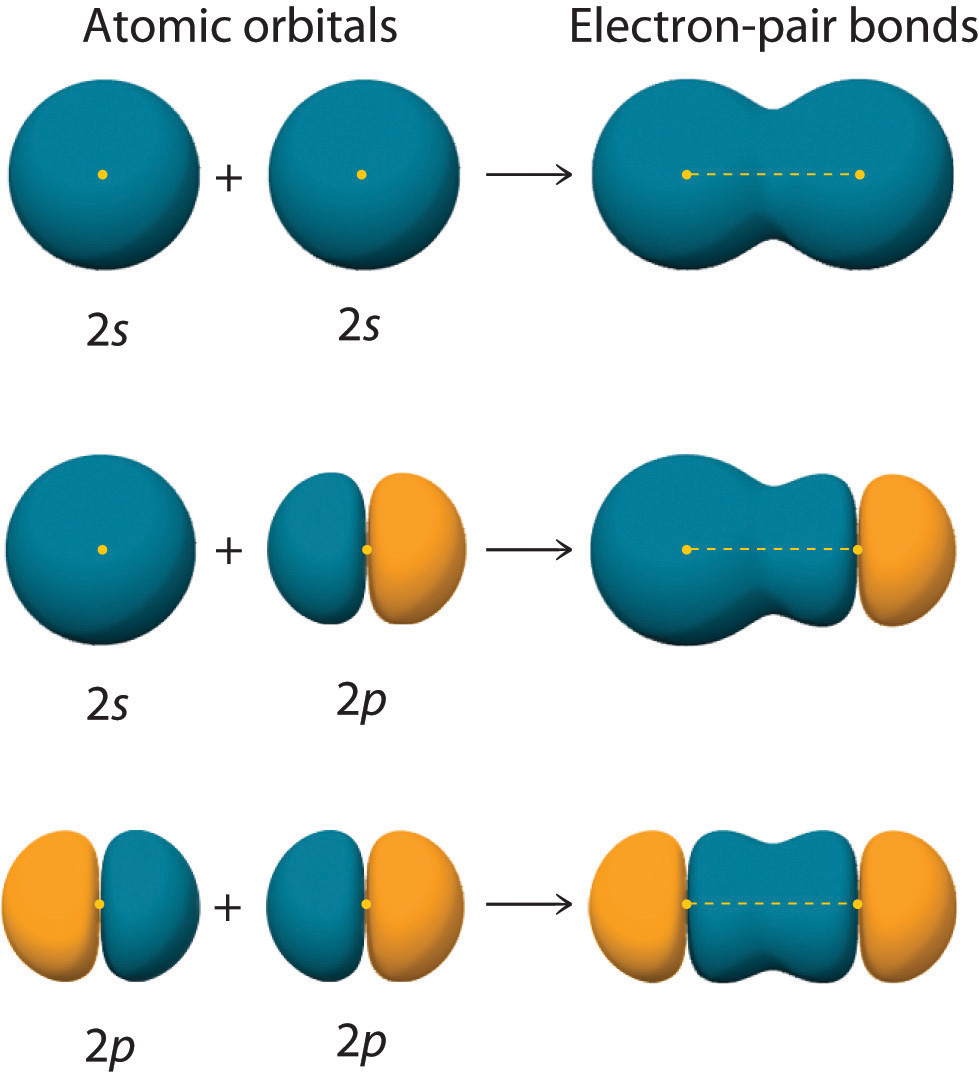
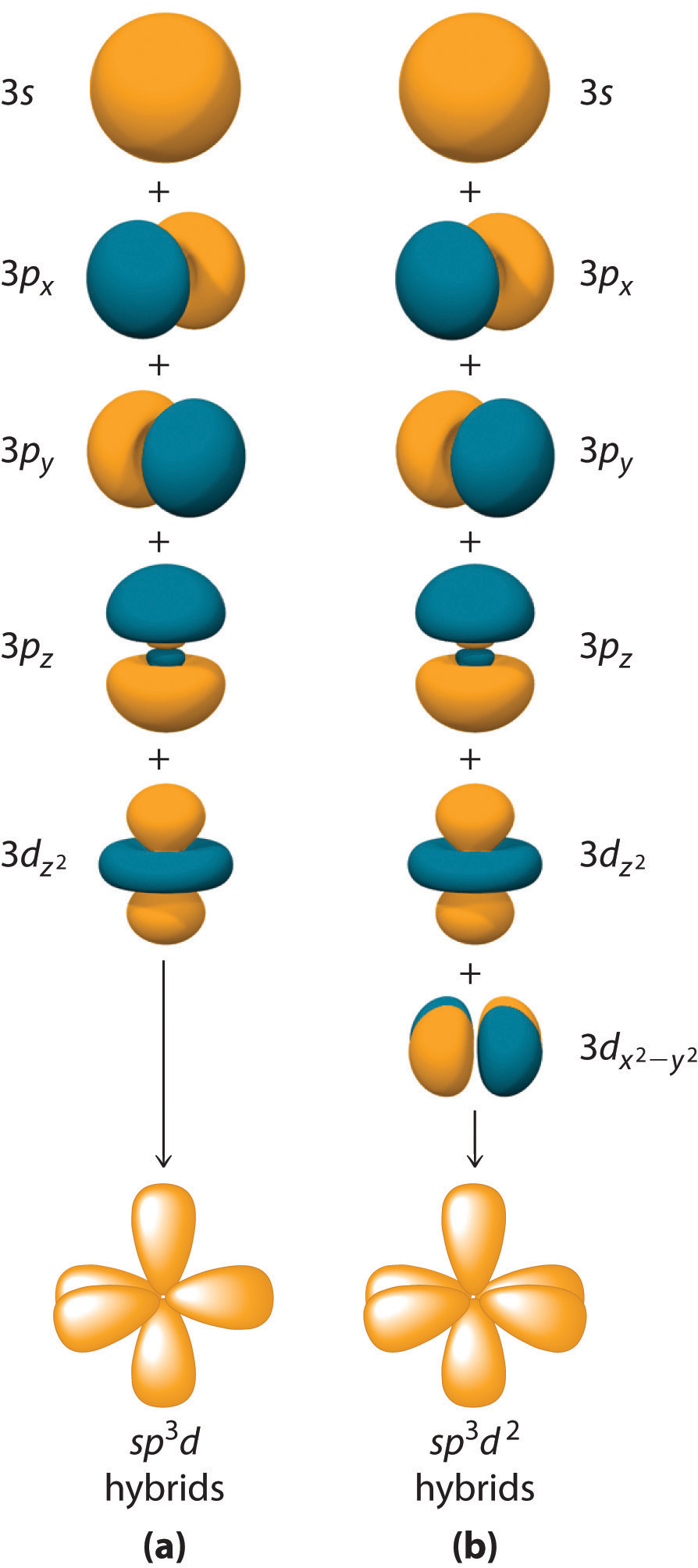
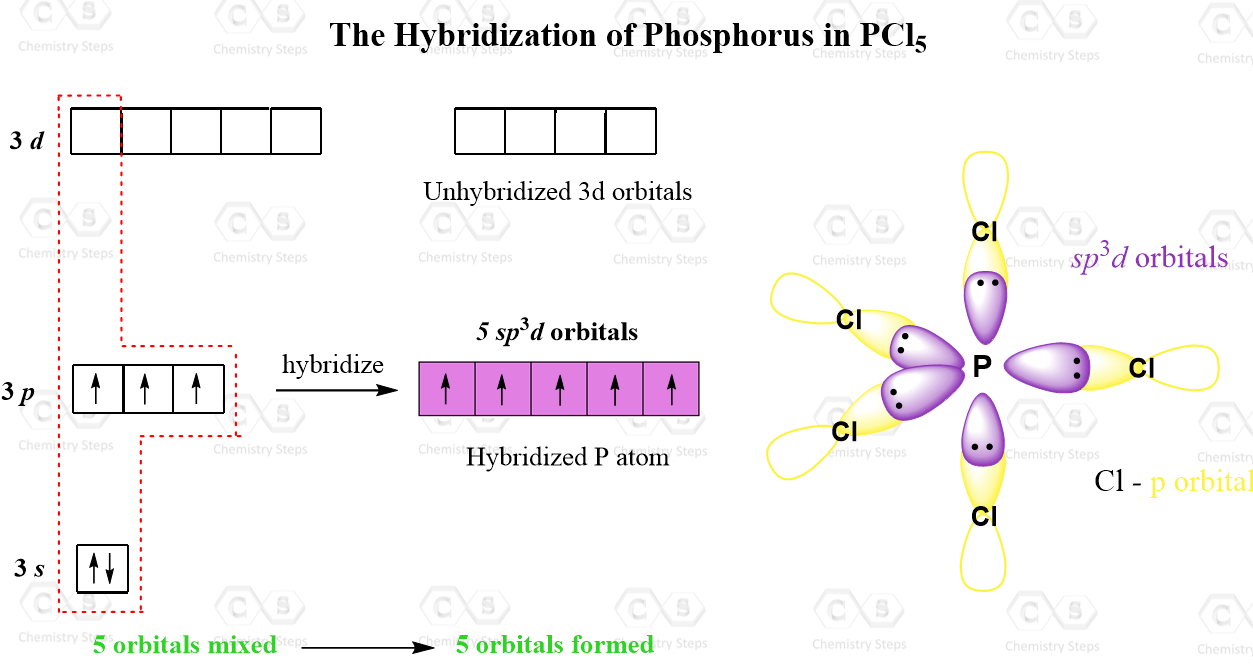
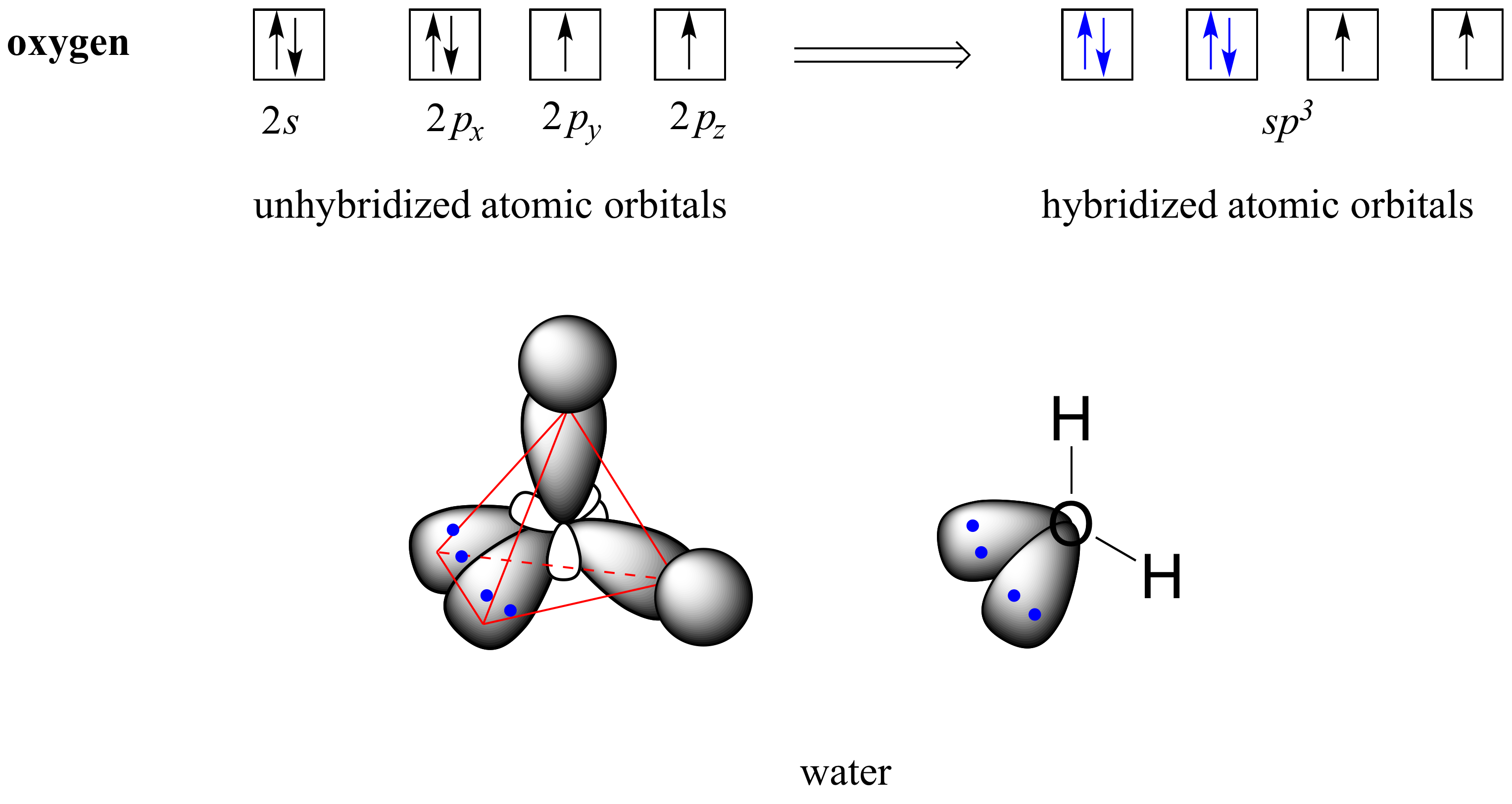
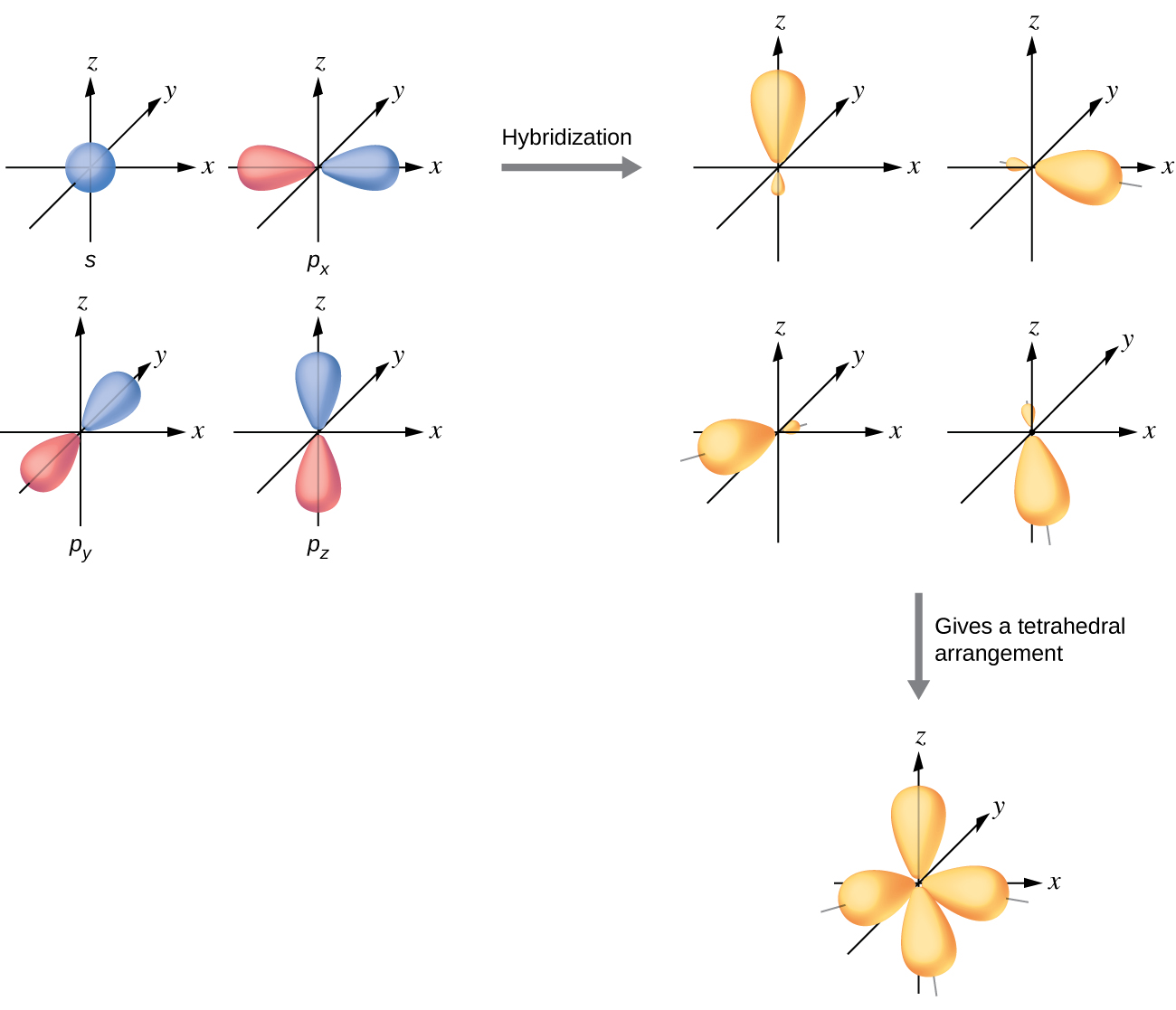
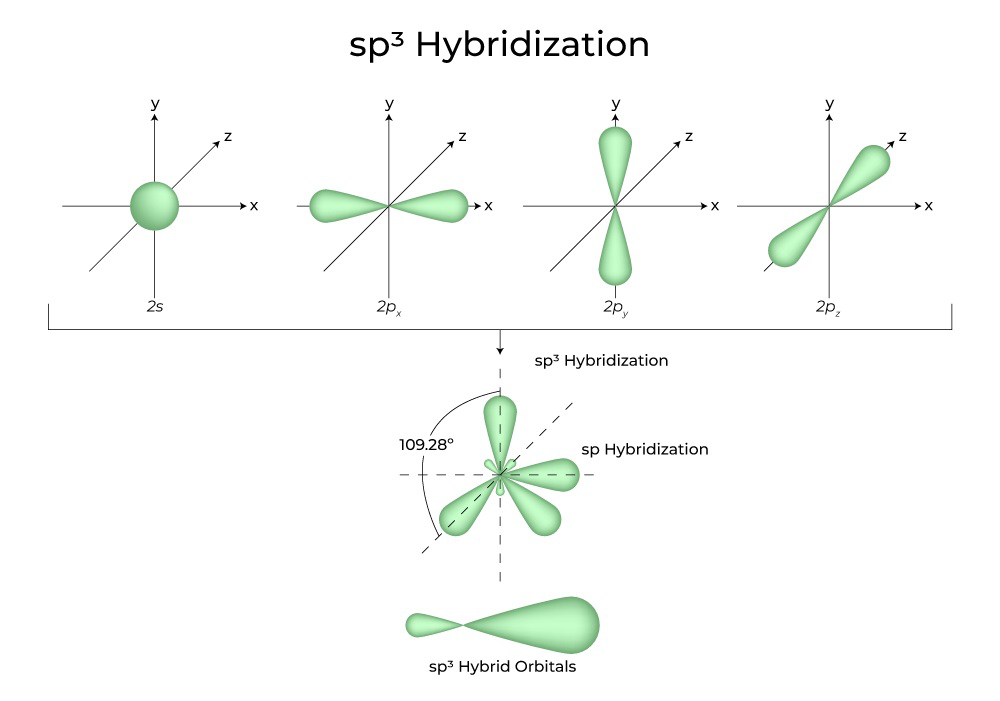
How To Draw Hybridization Orbitals
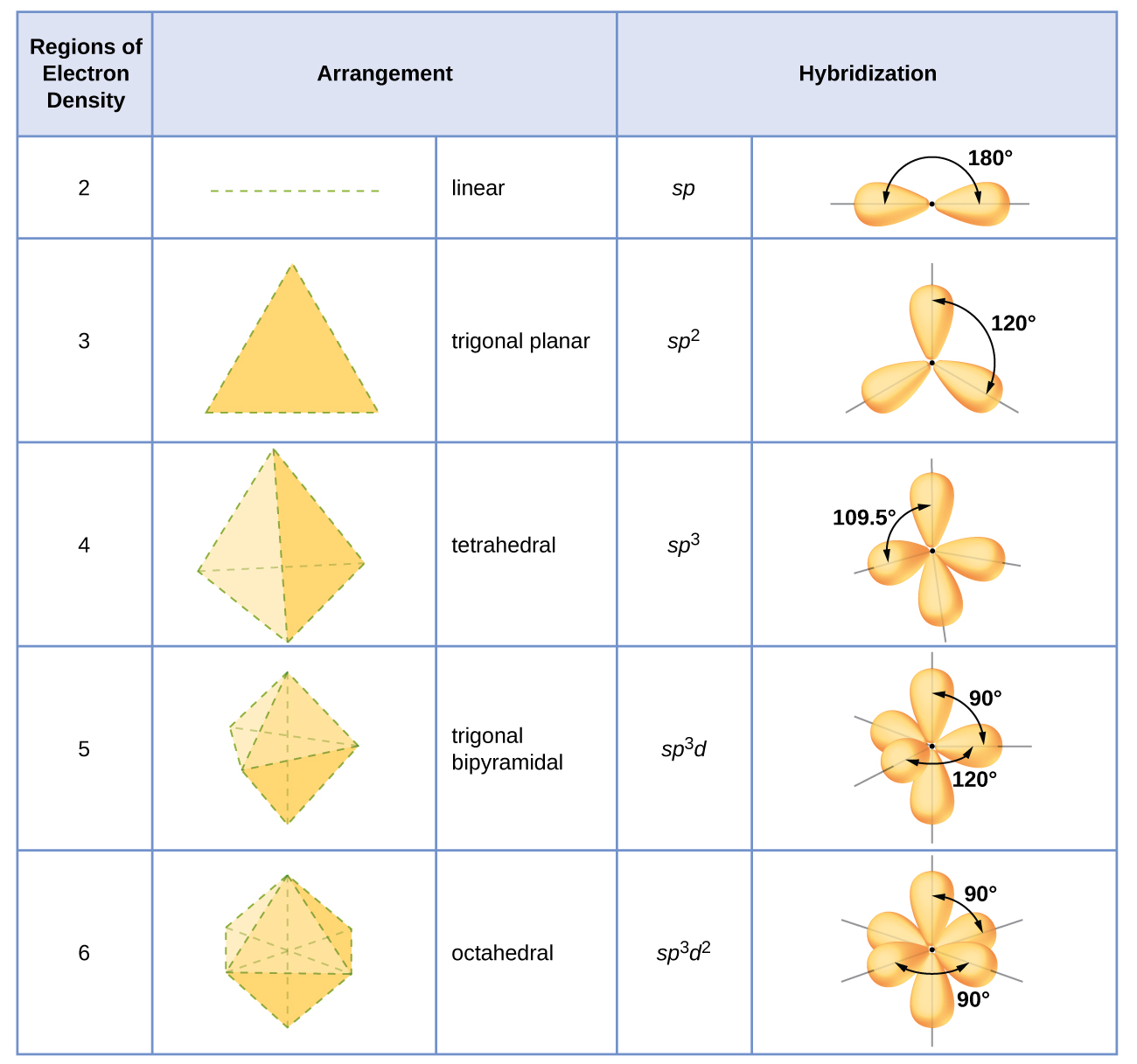
How To Draw Hybridization Orbitals - Note that each sp orbital contains one lobe that is significantly larger than the other. Draw orbital box diagrams showing how combinations of an atomic s orbital and various numbers of p orbitals create sp, sp 2, and sp 3 hybrid orbitals. Web steps to draw hybridized orbitals: Web directory of chem help asap videos: This makes it a challenge to draw, but i will show you the strategies in the video.in particular, you need to sho. Merge the orbitals depending on the type of hybridization: The nucleus resides just inside the minor lobe of. Hybridization of an s orbital (blue) and a p orbital (red) of the same atom produces two sp hybrid orbitals (purple). It discusses how to determine the number of sigma and pi bonds in a mol. From the valence electron configuration of the central atom and the number of electron pairs, determine the hybridization.
Web this video explains how to decide what the hybridization around an atom will be and also discusses the bonding picture of ethylene (ethene). In it, the 2s orbitals and two of the 2p orbitals hybridize to form three sp orbitals, each consisting of 67% p and 33% s character. Resonance structures, on the other hand, are different ways of. Place the total number of electrons around the central atom in the hybrid orbitals and describe the bonding. In beh 2, we can generate two equivalent orbitals by combining the 2s orbital of beryllium and any one of the three degenerate 2p orbitals. It discusses how to determine the number of sigma and pi bonds in a mol. The frontal lobes align themselves in the trigonal planar structure, pointing to the corners of a triangle in order. Unhybridized orbitals overlap to form π bonds. Hybrid orbitals overlap to form σ bonds. Web the carbon atoms of c2h2 are sp hybridized.
The formation of sp hybrid orbitals. The nucleus resides just inside the minor lobe of. This type of hybridization is required whenever an atom is surrounded by three groups of electrons. Web each individual hybrid orbital is a combination of multiple atomic orbitals and has different s and p character affecting their shape, length, and acidic properties. It discusses how to determine the number of sigma and pi bonds in a mol. Determine the steric number and hybridization of the center atom. Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character. In sp² hybridization, one s orbital and two p orbitals hybridize to form three sp² orbitals, each consisting of 33% s character and 67% p character. Web this organic chemistry video tutorial explains the hybridization of atomic orbitals. Note that each sp orbital contains one lobe that is significantly larger than the other.
Hybridization of Atomic Orbitals Chemistry Steps
Web figure 11.3.1 11.3. Web the nitrogen atom of ammonia (nh3) is sp3 hybridized. The frontal lobes align themselves in the trigonal planar structure, pointing to the corners of a triangle in order. In it, the 2s orbitals and two of the 2p orbitals hybridize to form three sp orbitals, each consisting of 67% p and 33% s character. It.
Orbital Hybridization "Cheat Sheet"? + Example
Determine the steric number and hybridization of the center atom. In this video, we use both of these methods to determine the hybridizations of atoms in various organic molecules. Web sp 2 hybridization. Draw orbital box diagrams showing how combinations of an atomic s orbital and various numbers of p orbitals create sp, sp 2, and sp 3 hybrid orbitals..
Hybridization of Atomic Orbitals Sigma & Pi Bonds Sp Sp2 Sp3 YouTube
The formation of sp hybrid orbitals. The frontal lobes align themselves in the trigonal planar structure, pointing to the corners of a triangle in order. Web steps to draw hybridized orbitals: In it, the 2s orbitals and two of the 2p orbitals hybridize to form three sp orbitals, each consisting of 67% p and 33% s character. Web the nitrogen.
Chapter 6.2 Hybrid Orbitals Chemistry LibreTexts
There are two oxygen atoms bonded to the center carbon atom and there are zero lone pairs around the carbon center. Web this video explains how to decide what the hybridization around an atom will be and also discusses the bonding picture of ethylene (ethene). Unhybridized orbitals overlap to form π bonds. Web directory of chem help asap videos: In.
Localized Bonding and Hybrid Atomic Orbitals
Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character. Web steps to draw hybridized orbitals: It discusses how to determine the number of sigma and pi bonds in a mol. The formation of sp hybrid orbitals. Web the carbon atoms of c2h2.
Hybridization of Atomic Orbitals Chemistry Steps
Merge the orbitals depending on the type of hybridization: Web figure 11.3.1 11.3. Hybridization of an s orbital (blue) and a p orbital (red) of the same atom produces two sp hybrid orbitals (purple). Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p.
2.2 Hybrid orbitals Chemistry LibreTexts
Hybrid orbitals overlap to form σ bonds. Determine the steric number and hybridization of the center atom. It discusses how to determine the number of sigma and pi bonds in a mol. By taking the sum and the difference of be 2s and 2p z atomic orbitals, for example, we produce two new orbitals with major and minor lobes oriented..
8.2 Hybrid Atomic Orbitals Chemistry
Figure 8.11 this alternate way of drawing the trigonal planar sp 2 hybrid orbitals is sometimes used in more crowded figures. It discusses how to determine the number of sigma and pi bonds in a mol. Hybrid orbitals overlap to form σ bonds. Web this organic chemistry video tutorial provides a basic introduction into valence bond theory and hybrid atomic.
Sp Orbitals
Draw orbital box diagrams showing how combinations of an atomic s orbital and various numbers of p orbitals create sp, sp 2, and sp 3 hybrid orbitals. Determine the steric number and hybridization of the center atom. The set of two sp orbitals are oriented at 180°, which is consistent with the. This makes it a challenge to draw, but.
8.2 Hybrid Atomic Orbitals Chemistry LibreTexts
Web hybridization of s and p orbitals. Web directory of chem help asap videos: In beh 2, we can generate two equivalent orbitals by combining the 2s orbital of beryllium and any one of the three degenerate 2p orbitals. Sp 2 hybridization can explain the trigonal planar structure of molecules. Figure 8.11 this alternate way of drawing the trigonal planar.
Web The Nitrogen Atom Of Ammonia (Nh3) Is Sp3 Hybridized.
Web figure 11.3.1 11.3. Note that each sp orbital contains one lobe that is significantly larger than the other. This type of hybridization is required whenever an atom is surrounded by two groups of electrons. Web directory of chem help asap videos:
Taking The Sum And Difference Of An Ns And An Np Atomic Orbital Where N = 2 Gives Two Equivalent Sp Hybrid Orbitals Oriented At 180° To Each Other.
In sp² hybridization, one s orbital and two p orbitals hybridize to form three sp² orbitals, each consisting of 33% s character and 67% p character. Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character. Web this organic chemistry video tutorial provides a basic introduction into valence bond theory and hybrid atomic orbitals. The frontal lobes align themselves in the trigonal planar structure, pointing to the corners of a triangle in order.
It Explains How To Find The Hybridi.
Web steps to draw hybridized orbitals: Place the total number of electrons around the central atom in the hybrid orbitals and describe the bonding. Sp 2 hybridization can explain the trigonal planar structure of molecules. The set of two sp orbitals are oriented at 180°, which is consistent with the.
From The Valence Electron Configuration Of The Central Atom And The Number Of Electron Pairs, Determine The Hybridization.
There are two oxygen atoms bonded to the center carbon atom and there are zero lone pairs around the carbon center. Merge the orbitals depending on the type of hybridization: By taking the sum and the difference of be 2s and 2p z atomic orbitals, for example, we produce two new orbitals with major and minor lobes oriented. Web this organic chemistry video tutorial explains the hybridization of atomic orbitals.